 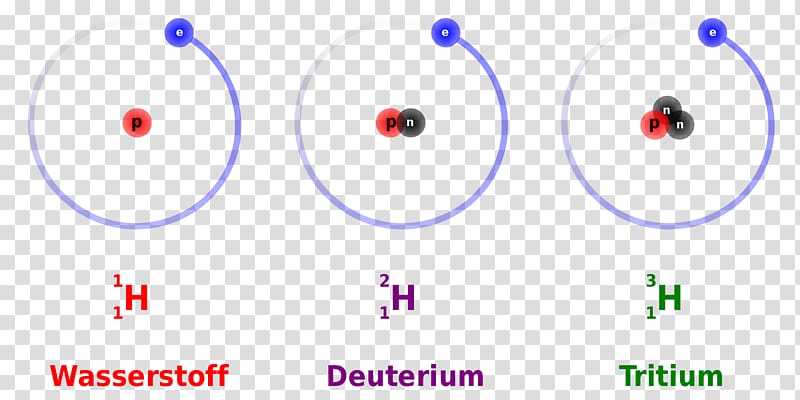
Some synthetic isotopes are much heavier than naturally occurring isotopes. Some potential applications of tritium include: In the research world, deuterium is used as a probe to study the structure and dynamics of molecules and to study the properties of neutron stars.Deuterium is also used as a tracer in biological research to study protein folding and metabolism.In the commercial world, deuterium is used as a tracer in chemical processes and as a moderator in nuclear reactors.Deuterium has a wide range of applications in both the commercial and research worlds.Deuterium and tritium are both radioactive, meaning they emit radiation. These isotopes are unstable because they have too few or too many neutrons, respectively, in their nuclei. There are three unstable isotopes of hydrogen: hydrogen-2 (deuterium), hydrogen-3 (tritium), and hydrogen-4 (quadrium). Hydrogen-2 has a proton and a pair of neutrons, and hydrogen-3 has a proton and a pair of protons. 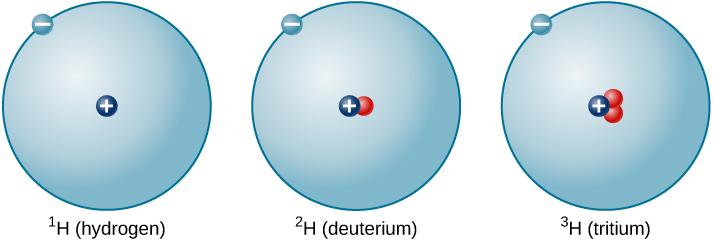
Hydrogen-1 is the most common, and it has a proton and a neutron. The isotopes of hydrogen are hydrogen-1, hydrogen-2, and hydrogen-3. Deuterium and tritium are also radioactive, with half-lives of 12.3 years and 12.5 years, respectively. Hydrogen-1 is a gas, while deuterium and tritium are solids at room temperature. Read more about how to correctly acknowledge RSC content.The isotopes of hydrogen have different physical properties. Permission is not required) please go to the Copyright If you want to reproduce the wholeĪrticle in a third-party commercial publication (excluding your thesis/dissertation for which If you are the author of this article, you do not need to request permission to reproduce figuresĪnd diagrams provided correct acknowledgement is given. Provided correct acknowledgement is given. If you are an author contributing to an RSC publication, you do not need to request permission Please go to the Copyright Clearance Center request page. To request permission to reproduce material from this article in a commercial publication, Provided that the correct acknowledgement is given and it is not used for commercial purposes. This article in other publications, without requesting further permission from the RSC, Hernández,Ĭreative Commons Attribution-NonCommercial 3.0 Unported Licence. 
Molecular hydrogen isotope separation by a graphdiyne membrane: a quantum-mechanical studyĮ. Permeances at these temperatures are below recommended values for practical applications, however, at slightly higher temperatures (77 K) they become acceptable while the selectivities preserve promising values, particularly for the separation of tritium. Finally, D 2/H 2 and T 2/H 2 selectivities are found to reach maximum values of about 6 and 21 at ≈50 and 45 K, respectively, a feature due to a balance between zero-point energy and tunneling effects in the transport dynamics. Moreover, it is found that a simple 1D model-using a zero-point energy-corrected interaction potential-provides an excellent agreement with the 3D probailities for perpendicular incidence conditions. The effect of the different incidence directions on the probabilities is analyzed in detail and it is concluded that restricting the simulations to a perpendicular incidence leads to reasonable results. The quantum dynamics of the molecules impacting on the membrane along a complete set of incidence directions have been rigorously addressed by means of wave packet calculations in the 3D space, which have allowed us to obtain transmission probabilities and, in turn, permeances, as the thermal average of the molecular flux per unit pressure. A reliable improved Lennard-Jones force field, optimized on accurate ab initio calculations, has been built to describe the molecule–membrane interaction, where the molecule is treated as a pseudoatom. In this work we study the separation of hydrogen isotopes in their transport through a GDY membrane due to mass-dependent quantum effects that are enhanced by the confinement provided by its intrinsic sub-nanometric pores. One of the most challenging goals is the separation of deuterium (D 2) and tritium (T 2) from a mixture with the most abundant hydrogen isotope, H 2, an achievement that would be of great value for a number of industrial and scientific applications. Graphdiyne (GDY) has emerged as a very promising two-dimensional (2D) membrane for gas separation technologies. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
